In collaboration with WHO and CDC, researchers at LSHTM have developed two vaccine schedule decision support models.
The first evaluates the potential health benefits and side-effects of rotavirus vaccination for schedules with and without age restrictions, and the second evaluates alternative Hib conjugate vaccine schedules. Both models track a cohort of children from birth to 5 years of age and generate estimates of the direct effectiveness of vaccination. These are calculated by applying estimates of vaccine efficacy (which vary by dose and time since vaccination) and coverage (which vary by age) to age-specific estimates of disease mortality. An important feature of the model is that very fine age intervals (weekly up to 1 year, and monthly thereafter) are used for the distributions of age at vaccination and at disease or death.
This allows for more precise estimates of the direct effects of different schedules than would otherwise be possible.
Each vaccine is currently evaluated independently. In reality many vaccines are administered at the same age, and to help answer questions about combined schedules future analyses will need to address this point.
Both models are developed in Excel to facilitate ease of use by national teams responsible for informing National Immunization Technical Advisory Committees (NITAGs). Countries interested in populating the model with local estimates, and evaluating the optimal schedule for their context, should contact Dr Ana Maria Henao Restrepo (henaorestrepoa@who.int) for further information.
Rotavirus vaccine risk-benefit model
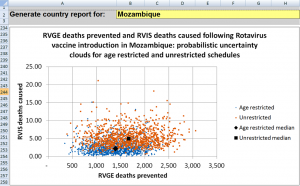 This model estimates numbers of deaths prevented by rotavirus vaccination and the numbers of excess intussusceptions deaths that might be caused by the vaccine. Outcomes can be assessed both with and without the manufacturer’s recommended age restrictions. The model includes options for scenario analysis and probabilistic sensitivity analysis.
This model estimates numbers of deaths prevented by rotavirus vaccination and the numbers of excess intussusceptions deaths that might be caused by the vaccine. Outcomes can be assessed both with and without the manufacturer’s recommended age restrictions. The model includes options for scenario analysis and probabilistic sensitivity analysis.
Hib vaccine schedules model
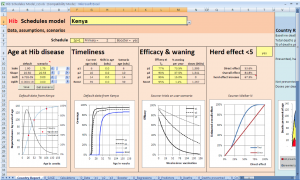 This model evaluates the potential numbers of deaths prevented by different Hib conjugate vaccination schedules. Of particular interest is the comparison of 2p+1 (two primary doses and a booster) and 3p+0 (three primary doses without a booster). The model includes options for scenario analysis, including the sensitivity of the results to some of the key drivers (e.g. the rate at which direct vaccine protection wanes with time since vaccination).
This model evaluates the potential numbers of deaths prevented by different Hib conjugate vaccination schedules. Of particular interest is the comparison of 2p+1 (two primary doses and a booster) and 3p+0 (three primary doses without a booster). The model includes options for scenario analysis, including the sensitivity of the results to some of the key drivers (e.g. the rate at which direct vaccine protection wanes with time since vaccination).
